INVESTOR INFORMATION
The Only Cell Therapy for Chronic Limb-Threatening Ischemia Available to US Patients Today
Hemostemix Inc. is a clinical-stage regenerative medicine company advancing ACP-01, an investigational autologous stem cell therapy derived from a patient's own blood, across six ischemic indications with active commercial access in the United States, Canada, and The Bahamas.
TSXV: HEM · OTCQB: HMTXF · FSE: 2VF0
THE PROBLEM
A Crisis Medicine Hasn't Solved
Every year, approximately 150,000 Americans lose a limb to chronic limb-threatening ischemia, a severe vascular disease that cuts off blood flow to the extremities until tissue dies. These are called no-option patients: people who have exhausted surgical and endovascular intervention and have nowhere left to turn.
Their prognosis without a new option is stark. A 50% five-year mortality rate. A 37% one-year amputation-free survival rate. Chronic pain, progressive disability, and decline.
 Cell therapy has long promised a solution for these patients. Four companies are now meaningfully in the race. Only one is treating patients in the United States today.
Cell therapy has long promised a solution for these patients. Four companies are now meaningfully in the race. Only one is treating patients in the United States today.
Cell therapy has long promised a solution for these patients. Four companies are now meaningfully in the race. Only one is treating patients in the United States today.
THE OPPORTUNITY
A Platform - Not A Single Product
ACP-01 targets six distinct ischemic and vascular conditions that share a common underlying mechanism: insufficient microvascular perfusion. A single basket IND filing unlocks a combined market estimated in the tens of millions of patients.
Patient population figures reflect published epidemiological estimates and are provided for market context only. They do not represent projected revenues or financial performance.
THE PLATFORM
ACP-01: A Different Kind Of Cell Therapy
Most cell therapies in development require invasive collection procedures: bone marrow aspiration, liposuction under general anesthesia, surgical harvest. These procedures are high-risk for the exact patients who need treatment most: elderly, diabetic, and medically fragile individuals.
ACP-01 starts with a blood draw. A standard peripheral venipuncture. The patient's CD34+ angiogenic cell precursors are isolated, expanded over seven days in a cGMP-compliant facility, and administered via outpatient intramuscular injection.
No surgery. No general anesthesia. No hospital admission.
7
Completed clinical studies
498
Subjects studied
11
Peer-reviewed publications
4.5 yr
Max published follow-up
90+
Patents protecting the platform
0
SAEs attributed to ACP-01
CLINICAL EVIDENCE
The Most Published Cell Therapy In This Space
ACP-01 has been evaluated in seven completed clinical studies across 498 subjects, with results published in 11 peer-reviewed journals -- more human evidence than any other cell therapy competing in CLTI. The published data are consistent across studies and follow-up periods extending to 4.5 years.
83% limb salvage
In treated CLTI cohorts followed to 4.5 years. Published across multiple study cohorts.
99.7% ulcer reduction
From 146 mm² to 0.48 mm² at three months. Henderson et al., 2024.
0% mortality
In CLTI cohorts through the published follow-up period of 4.5 years.
Zero SAEs
No serious adverse events attributed to ACP-01 across all 498 subjects in published studies.
These results represent published investigational findings. ACP-01 has not received FDA or Health Canada marketing approval. Individual outcomes in future studies may vary. Data sourced from peer-reviewed publications available at hemostemix.com/scientific-publications.
COMPETITIVE POSITION
Where ACP-01 Stands In The Field
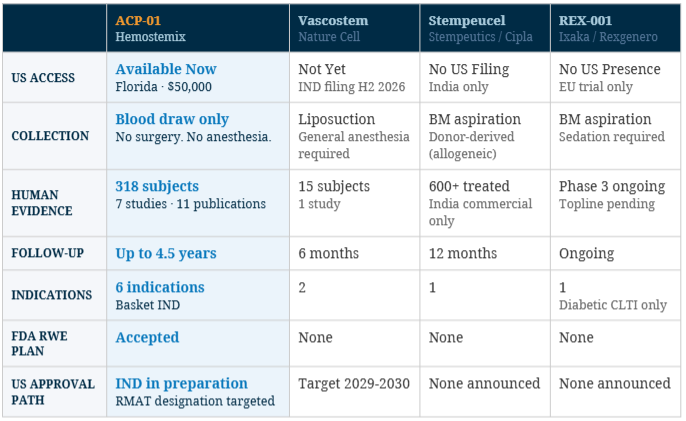
Four companies are competing in CLTI cell therapy as of April 2026. The landscape is early, the window is open, and the first-mover advantage is measurable.
|
|
ACP-01 Hemostemix |
Vascostem Nature Cell |
Stempeucel Stempeutics / Cipla |
REX-001 Ixaka / Rexgenero |
|
US ACCESS |
Available Now Florida |
Not Yet IND filing H2 2026 |
No US Filing India only |
No US Presence EU trial only |
|
COLLECTION |
Blood draw only No surgery. No anesthesia. |
Liposuction General anesthesia required |
BM aspiration Donor-derived (allogeneic) |
BM aspiration Sedation required |
|
HUMAN EVIDENCE |
498 subjects 7 studies · 11 publications |
15 subjects 1 study |
600+ treated India commercial only |
Phase 3 ongoing Topline pending |
|
FOLLOW-UP |
Up to 4.5 years |
6 months |
12 months |
Ongoing |
|
INDICATIONS |
6 indications Basket IND |
2 |
1 |
1 Diabetic CLTI only |
|
FDA RWE PLAN |
Accepted |
None |
None |
None |
|
US APPROVAL PATH |
IND in preparation RMAT designation targeted |
Target 2029-2030 |
None announced |
None announced |
No product in this landscape holds FDA or EMA marketing authorization for CLTI as of April 2026. Competitive data sourced from company press releases, ClinicalTrials.gov, and peer-reviewed publications. Forward-looking statements reflect publicly available information and are not guarantees of future outcomes.
SAFETY PROFILE
Autologous vs. Allogeneic:
Why The Source Of The Cells Matters
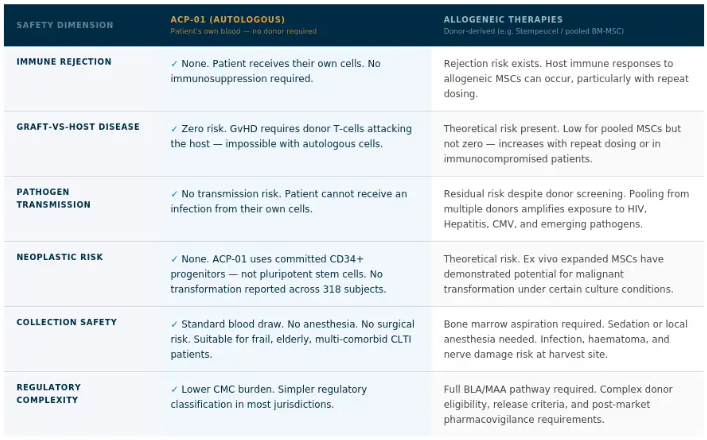
One of the most consequential distinctions in cell therapy is whether the cells come from the patient or from a donor. ACP-01 is autologous, derived entirely from the patient's own blood. This eliminates every category of donor-derived risk by definition. There is no donor. There is no foreign material. There is no immune mismatch.
|
Safety Dimension |
ACP-01 (Autologous) Patient's own blood |
Allogeneic Therapies Donor-derived (e.g. Stempeucel) |
|
IMMUNE REJECTION |
None. Patient receives their own cells. No immunosuppression required. |
Rejection risk exists. Host immune responses to allogeneic MSCs can occur, particularly with repeat dosing. |
|
GRAFT-VS-HOST DISEASE |
Zero risk. GvHD requires donor T-cells attacking the host -- impossible with autologous cells. |
Theoretical risk present. Low for pooled MSCs, but not zero -- increases with repeat dosing or in immunocompromised patients. |
|
PATHOGEN TRANSMISSION |
No transmission risk. Patient cannot receive an infection from their own cells. |
Residual risk despite donor screening. Pooling from multiple donors amplifies exposure to HIV, Hepatitis, CMV, and emerging pathogens. |
|
NEOPLASTIC RISK |
None. ACP-01 uses committed CD34+ progenitors -- not pluripotent stem cells. No transformation reported in 318 subjects. |
Theoretical risk. Ex vivo expanded MSCs have demonstrated potential for malignant transformation under certain culture conditions. |
|
COLLECTION SAFETY |
Standard blood draw. No anesthesia. No surgical risk. Suitable for frail, elderly, multi-comorbid patients. |
Bone marrow aspiration required. Sedation or local anesthesia needed. Infection, haematoma, and nerve damage risk at harvest site. |
|
REGULATORY COMPLEXITY |
Lower CMC burden. Simpler regulatory classification in most jurisdictions. |
Full BLA/MAA pathway required. Complex donor eligibility, release criteria, and post-market pharmacovigilance requirements. |
Safety profile based on published peer-reviewed literature. No serious adverse events have been attributed to ACP-01 across seven clinical studies and 318 subjects. ACP-01 remains investigational in all indications. References: Henderson et al. 2024; Gupta et al. Stem Cell Research & Therapy 2023; Lazarus HM et al. Blood 2005.
Autologous vs. Allogeneic:
Why The Source Of The Cells Matters
One of the most consequential distinctions in cell therapy is whether the cells come from the patient or from a donor. ACP-01 is autologous, derived entirely from the patient's own blood. This eliminates every category of donor-derived risk by definition. There is no donor. There is no foreign material. There is no immune mismatch.
SAFETY PROFILE
Active In The US Market Right Now
Florida, today.
Under Florida Senate Bill 1768, effective July 1, 2025, physicians may offer autologous stem cell therapies within their scope of practice, provided informed consent and cGMP-compliant manufacturing are obtained. ACP-01 is lawfully available in Florida with every procedure conducted under IRB oversight.
This is not a workaround. It is a regulatory strategy. Every patient treated generates real-world evidence under a framework the FDA has explicitly accepted. That evidence flows directly into the basket IND filing.
ACP-01 is positioned well below the $300,000-$500,000 benchmarks set by approved cell and gene therapies in adjacent indications. Treatment is also available in Canada and The Bahamas.
The basket IND preparation is ongoing following the January 2026 FDA pre-IND meeting. RMAT designation is being targeted, which would provide intensive FDA guidance, rolling review, and potentially accelerated approval. Hemostemix is the only company in this space actively building FDA-accepted RWE under IRB oversight. Competitors who have not started this process will be years behind when they eventually seek a similar pathway.
All regulatory and commercial milestones are subject to FDA oversight and applicable jurisdictional requirements. Forward-looking statements reflect current plans and are not guarantees of future outcomes.
WHY HEMOSTEMIX
Six Reasons The Competitive Window Belongs To ACP-01
First-mover commercial access
ACP-01 is the only cell therapy for CLTI available to US patients today. No competitor can make this claim.
Only non-invasive collection
A blood draw vs. bone marrow aspiration or liposuction. The only method appropriate for the full breadth of the no-option CLTI population.
FDA-accepted RWE strategy
The only company in this space building FDA-accepted real-world evidence under IRB oversight through active US commercial sites.
Most published evidence in class
7 studies. 498 subjects. 11 peer-reviewed publications. 4.5 years of follow-up. More human evidence than every CLTI competitor combined.
Six-indication basket IND
A single regulatory filing unlocks a combined addressable market across cardiac, vascular, and neurological ischemic disease.
Patient-accessible pricing
$50,000 per treatment. A fraction of the cost of approved cell and gene therapies in adjacent indications.
ACP-01 remains investigational in all indications. All forward-looking statements are subject to risk and uncertainty. Investors should review all public disclosures before making investment decisions.
CAPITAL STRUCTURE
Corporate Information
HEM
HMTXF
2VF0
Frankfurt Stock Exchange
The company maintains regulatory filings and public disclosures in accordance with applicable exchange requirements. Investors may access financial statements, management discussion and analysis, corporate governance documentation, and press releases via SEDAR+.
Contact Investor Relations
For institutional inquiries, partnership discussions, or investor communications, please contact:
Latest News
HEMOSTEMIX CLOSES $303,967 PRIVATE PLACEMENT AT $0.12 PER SHARE
Calgary, Alberta – March 19, 2026 – Hemostemix Inc. (TSXV: HEM | OTCQB: HMTXF | FSE: 2VF0) (the “...
Dr. Ernst von Schwarz to Host Webinar: VesCell for Advanced Cardiac Conditions
May 5th, 12:00 p.m. Four published studies of 24, 41, 106, 54 subjects reviewed - treated for...
Hemostemix’s Insider Lead Order of $330,000 Private Placement
Calgary, Alberta, October 7, 2025 - Hemostemix (TSXV: HEM) (OTC: HMTXF) (FSE: 2VF0) ("Hemostemix"...
Annual General Meeting Materials
2024
2023
These results represent published investigational findings. ACP-01 has not received FDA or Health Canada marketing approval. Individual outcomes in future studies may vary. Data sourced from peer-reviewed publications available at hemostemix.com/scientific-publications.
Disclaimer

